
Display:Ĭhromium metal piece and flakes, stainless steel nuts and bolts, chrome-plated plumbing fixtures, crocoite PbCrO4, rubies and a ruby wand Al2O3♼r, and paints with chromium pigments – chrome yellow (PbCrO4) and chromium oxide green (Cr2O3)

Vanadium oxides are used as catalysts and as fixes for the dyeing of fabrics. Vanadium is used in alloys to increase strength, most commonly in steels, and in lighter titanium alloys for the aerospace industry. The name rionium, honouring del Rio, was also proposed but not adopted. Sefstrom named the element vanadium after the Norse goddess Vanadis, who amongst other things was the goddess of beauty, reflecting the range of beautiful colours of vanadium compounds, and because the symbol V was not in use.
In 1831, vanadium was rediscovered by Nils Gabriel Sefstrom, and del Rio’s work was verified by Friedrich Wohler. However, in 1805 it was claimed that del Rio had mistaken chromium for a new element, causing del Rio to retract his discovery.
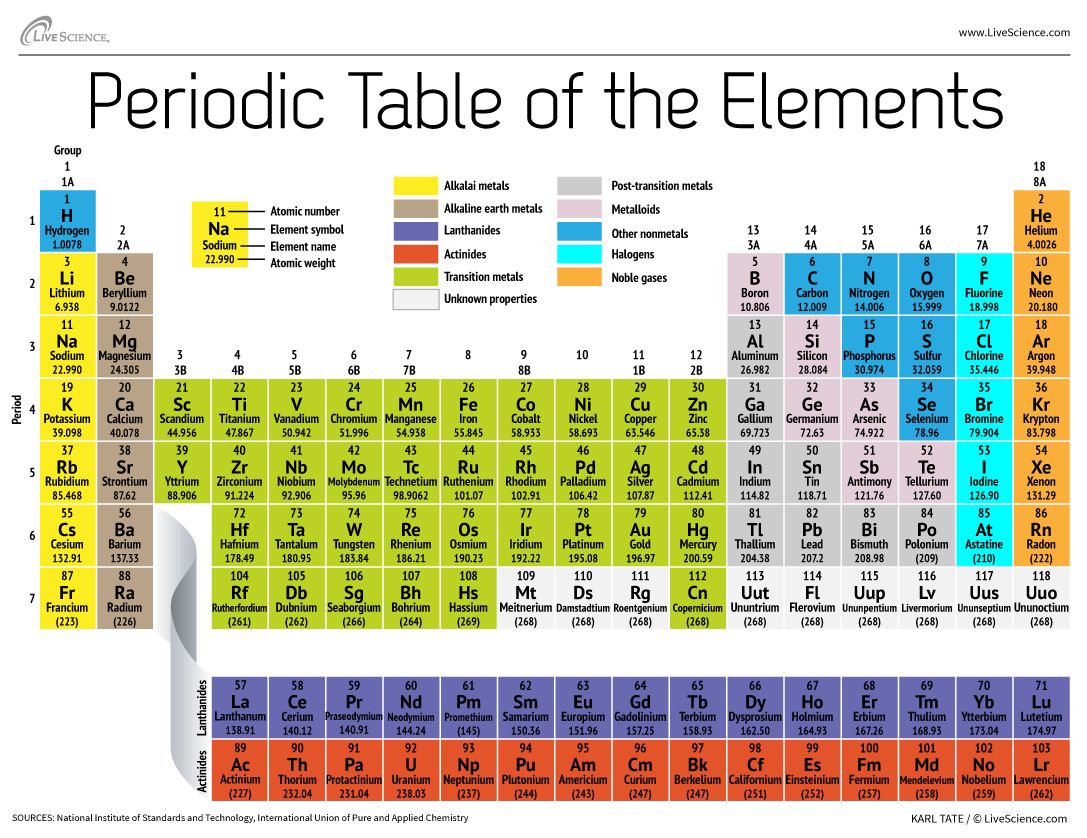
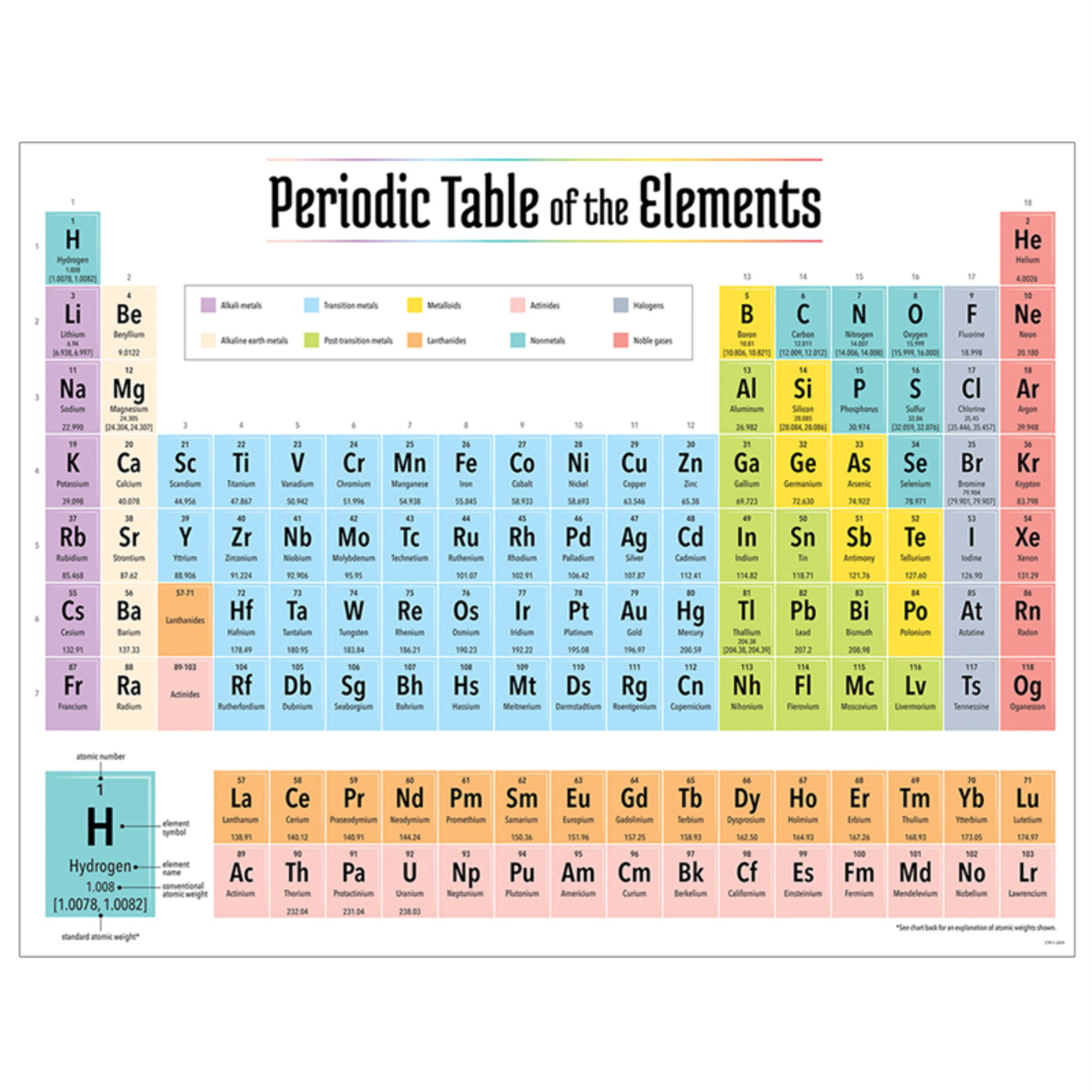
He then changed the name to erythronium (meaning red in Greek) as many vanadium salts turned red when heated. He named the element panchromium (meaning all-colours in Greek) as it produced salts of many different colours. In 1801, Andres Manuel del Rio discovered vanadium in an ore called brown lead (now called vanadinite). (In the modern periodic table, a group or family corresponds to one vertical column.Titanium metal pellet and rod, titanium oxides – TiO (beige), Ti 2O 3 (purple), TiO 2 (white, also known as titania), titania-containing sunscreen and white paint, and rutilated quartz (quartz contained rutile TiO 2) The periodic table allows chemists a shortcut by arranging typical elements according to their properties and putting the others into groups or families with similar chemical characteristics. Were it not for the simplification provided by this chart, students of chemistry would need to learn the properties of all 118 known elements. The term “periodic” is based on the discovery that elements show patterns in their chemical properties at certain regular intervals. Mendeleev left spaces for elements he expected to be discovered, and today’s periodic table contains 118 elements, starting with hydrogen and ending with oganesson, a chemical element first synthesized in 2002 at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, by a team of Russian and American scientists. Its story is over 200 years old, and throughout its history, it has been a subject for debate, dispute and alteration.Īttempts to classify elements and group them in ways that explained their behavior date back to the 1700s, but the first actual periodic table is generally credited to Dmitri Ivanovich Mendeleev, a Russian chemist who in 1869 arranged 63 known elements according to their increasing atomic weight. Go into any scientist’s office or lecture hall anywhere in the world and you are likely to see one. There is no more enduring reflection of science than the Periodic Table of Chemical Elements, which sheds light not only on the essence of chemistry but physics and biology as well.
